A first-in-human phase 1 trial has brought an intriguing new vision-restoration concept into the clinic: intravitreal photoswitch therapy for advanced retinitis pigmentosa (RP). Reported in Nature Medicine, the open-label ABACUS-1 study evaluated KIO-301, a small azobenzene-based molecule designed to confer light responsiveness to surviving retinal ganglion cells in eyes with end-stage photoreceptor loss. In a field dominated by gene-specific approaches, the appeal is obvious: this is a gene-agnostic strategy aimed at patients with profound vision loss, regardless of the underlying mutation.
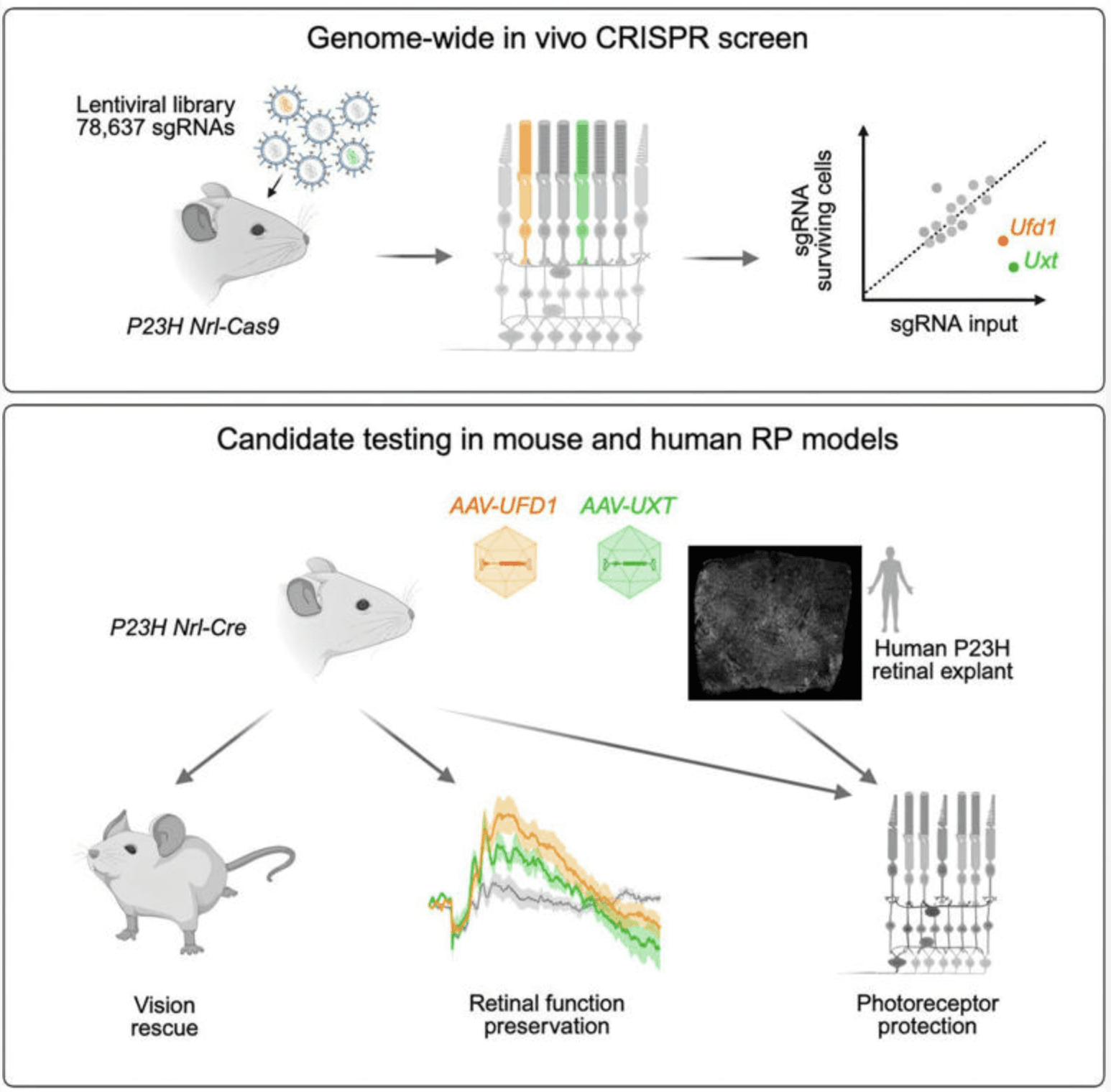
Researchers in the WashU Medicine Department of Ophthalmology & Visual Sciences have discovered key neuroprotective genes that could lead to the development of gene therapies to treat retinitis pigmentosa, an inherited form of retinal degeneration that causes blindness. The findings, published in Neuron, point to new therapeutic strategies to maintain retinal health and protect against degeneration.
"Previous studies in our laboratory have demonstrated the neuroprotective and retinoprotective actions of 670 nm light treatment in vitro and in vivo. The present study extends these findings to include protection against photoreceptor degeneration in P23H rats. The results of these studies further support the use of 670 nm light therapy in the treatment of retinal injury and degenerative retinal disease"
MCO-010, Nanoscope's proprietary multi-characteristic opsin (MCO), is delivered via a one-time, in-office intravitreal injection. After injection, MCO-010 activates highly dense bipolar retinal cells to become light sensitive, utilizing remaining visual circuitry following photoreceptor death. MCO-010 is designed to offer a distinct approach in that it does not require genetic testing, surgical intervention, or repeat dosing, making it applicable to a broad population of RP patients, while also fitting into existing retina office workflows.
New studies in rats suggest the drug reserpine, approved in 1955 for high blood pressure, might treat the blinding disease retinitis pigmentosa. No therapy exists for this rare inherited disease, which starts affecting vision from childhood. A report on the studies, conducted at the National Institutes of Health (NIH), published today in eLife.
“The discovery of reserpine’s effectiveness may greatly speed therapeutics for retinitis pigmentosa and many other inherited retinal dystrophies, which can be caused by one of more than a thousand possible mutations affecting more than 100 genes. Reserpine’s neuroprotective effect is independent of any specific underlying gene mutation,” said the study’s lead investigator, Anand Swaroop, Ph.D., senior investigator at NIH’s National Eye Institute.
"In a groundbreaking development for ophthalmology, researchers from KAIST have achieved the first successful induction of long-term neural regeneration in mammalian retinas, potentially paving the way to restore vision lost to degenerative retinal diseases."
A previously unreported population of human stem-like cells capable of regenerating retinal tissue and potentially promoting vision recovery has been identified.
A team led by Professor Jianzhong Su from Wenzhou Medical University, China, has identified human neural retinal stem-like cells (hNRSCs) with the capability to regenerate retinal tissue and the potential to assist in vision recovery.
The research team successfully induced retinal regeneration and vision recovery in a disease-model mouse by administering a compound that blocks the PROX1 (prospero homeobox 1) protein, which suppresses retinal regeneration. Furthermore, the effect lasted for more than six months.
Scientists say they have found a long–sought-after population of stem cells in the retina of human fetuses that could be used to develop therapies for one of the leading causes of blindness. The use of fetal tissue, a source of ethical debate and controversy in some countries, likely wouldn’t be necessary for an eventual therapy: Transplanting similar human cells generated in the lab into the eyes of mice with retinal disease protected the animals’ vision, the team reported this week in Science Translational Medicine.
“I see this as potentially a very interesting advancement of this field, where we are really in need of a regenerative treatment for retinal diseases,” says Anders Kvanta, a retinal specialist at the Karolinska Institute who was not involved in the work.
"This study evaluates the neuroprotective effects of voluntary exercise in a mouse model of RP and investigates the role of the adiponectin signaling pathway....These findings demonstrate that voluntary exercise enhances photoreceptor survival in RP, potentially via increased serum adiponectin levels, while elevated retinal adiponectin may represent a compensatory response to degeneration. This study highlights the therapeutic potential of exercise in RP and identifies adiponectin as a promising target for further investigation into neuroprotective mechanisms and treatments."